'Product Blog'
Silicon Based Electrolyte Solvents for Lithium Ion Batteries
Electrochemically and Thermally Stable Polysiloxane Electrolytes Improve the Long Time Operation of Lithium Ion Batteries
Electrolytic blend, which normally includes a lithium metal salt, an aprotic solvent and a redox shuttle, is one of the most economic and effective methods for the improvement of Li-ion battery performance.[1, 2] There are many types of electrolyte solvents, which are mainly characterized by conductivity, polarity (in order to dissolve the lithium salt), and thermal/ electrochemical stability with a wide potential range (0−5.0 V vs Li/Li+).[2]
A team from Argonne National Laboratory developed silicon-based electrolytes that have emerged as primary candidates for the development of lithium-ion battery technologies. Compared to the standard carbonate based electrolytes, silicon-based electrolytes are thermally and chemically stable, less flammable, and environmental friendly.[3]
|
|
(14-1925) ANL-1NM2 |
|
|
(14-1930) ANL-1NM3 |
ANL- 1NM2 (14-1930) having a high conductivity of 1.2 x 10–3 S cm–1 at 25°C (doped with 1.0 M LiTFSI) has better electrochemical stability than its germanium and carbon analogues.[4] However, short ethylene oxide chained silane restricts the choice of lithium salts. The best soluble lithium salt in ANL- 1NM2 is LiTFSI which can be dissolved up to 1.2 M.[4]
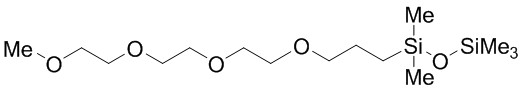
ANL-1NM3 (14-1930) attracts attention due to its high conductivity (0.8M LiBOB doped electrolyte: 1.18 x 10–3 S cm–1 at 25°C or 1.0 M LiTFSI doped electrolyte: 1.0 x 10–3 S cm–1 at 25°C) and low viscosity (h = 1.4 cP at 25°C).[5] Moreover, ANL-1NM3 (14-1930) electrolytes with a 0.8M LiBOB salt concentration are stable to 4.4 V[6], despite the fact that most oligoether linkages (CH2−CH2−O)n undergo oxidative decomposition above 4.0 V.[2]
|
|
(14-1943) ANL-2SM3 |
|
|
(14-1946) ANL-1S1M3 |
ANL-2SM3 (14-1943) also exhibits high electrochemical stability, high thermal stability, and low viscosity (h = 3.8 cP at 25°C). Disiloxane electrolytes doped with 0.8M LiBOB are stable to 4.7 V [7]. Compared to most of the siloxanes, ANL-2SM3 electrolyte solvent can dissolve more lithium salts such as LiBOB, LiPF6 (03-0325), LiBF4 (03-0325), and LiTFSI.
ANL-1S1M3 (14-1946) electrolyte solvent has fewer limitations towards the solvation of Li+ and can dissociate LiBOB, LiPF6 (03-0325), and LiTFSI. ANL-1S1M3 is non-hydrolyzable and less flammable than alkoxysilane counterparts [8]. The conductivity of 0.8M LiBOB in ANL-1S1M3 electrolyte is 1.29 x 10–3 S cm–1 at 25°C.
References:
- S. S. Zhang, J. Power Sources, 2006, 22, 1379.
- K. Xu, Chem. Rev., 2014, 114, 11503.
- S. S. Zhang, J. Power Sources, 2006, 22, 1379.
- L. Zhang et al. J. Mater. Chem., 2008, 18, 3713.
- Z. Chen et al. J. Phys. Chem. C, 2008, 112, 2210.
- K. Amine et al., Electrochem. Commun. 8 (2006) 429.
- Z. Zang et al, J. Power Sources, 2010, 195, 6062.
- L. Zhang et al, J. Mater. Chem., 2010, 20, 8224.
Products mentioned in this blog and related products:
|
1,4-Di-t-butyl-2,5-bis(2-methoxyethoxy)benzene, 99+% Redox shuttle ANL-RS2 |
|
|
6,7-Dimethoxy-1,1,4,4-tetramethyl-1,2,3,4-tetrahydronaphthalene, 99+% Redox shuttle ANL-RS21 |
|
|
2,2-Dimethyl-3,6,9-trioxa-2-siladecane, 99+% Electrolyte solvent ANL-1NM2 |
|
|
2,2-Dimethyl-3,6,9,12-tetroxa-2-silatridecane, 99+% Electrolyte solvent ANL-1NM3 |
|
|
2,2,4,4-Tetramethyl-3,8,11,14,17-pentaoxa-2,4-disilaoctadecane, 99+% Electrolyte solvent ANL-2SM3 |
|
|
2,2-Dimethyl-4,7,10,13-tetraoxa-2-silatetradecane, 99+% Electrolyte solid ANL-1S1M3 |
|
|
(2,5-Dimethoxy-1,4-phenylene)bis(di-i-propylphosphine oxide), 99+% Redox shuttle ANL-RS5 |
|
|
(2,5-Dimethoxy-1,4-phenylene)bis(diethylphosphine oxide), 99+% Redox shuttle ANL-RS51 |
|
|
2,5-Di-t-butyl-1,4-phenylene tetraethyl bis(phosphonate), 99+% Redox shuttle ANL-RS6 |
Visit the below link to view additional details on our website:
Materials for Battery Applications Redox Shuttles & Electrolyte Solvents