'Product Blog'
Bambus[6]Urils - An Anion Receptor with Exceptional Binding Properties
The macrocycle that demonstrates outstanding host-guest binding properties
Bambus[6]urils is related to the supramolecular cucurbit[n]urils group, where n glycoluril (![]() ) units are connected by 2n methylene bridges. Cucurbit[n]urils are the kind of molecular containers that are able to form stable complexes with various guests, including drug molecules, amino acids and peptides, saccharides, dyes, hydrocarbons, perfluorinated hydrocarbons, and even high molecular weight guests such as proteins. These molecules are also able to promote cucurbituril-catalyzed reactions [1].
) units are connected by 2n methylene bridges. Cucurbit[n]urils are the kind of molecular containers that are able to form stable complexes with various guests, including drug molecules, amino acids and peptides, saccharides, dyes, hydrocarbons, perfluorinated hydrocarbons, and even high molecular weight guests such as proteins. These molecules are also able to promote cucurbituril-catalyzed reactions [1].
Macrocycles of Bambus[6]urils, developed by Czech scientists in 2010 [2], may be represented as a class of neutral anion receptors. Since the structural and binding features of Bambus[6]uril [Fig. 1] differ significantly from those of the cucurbit[n]uril family, the authors proposed the descriptive name Bambus[6]uril for the macrocycle. The name is derived from the subfamily Bambusoideae, as the macrocycle cross sectional structure resembles part of the bamboo plant.
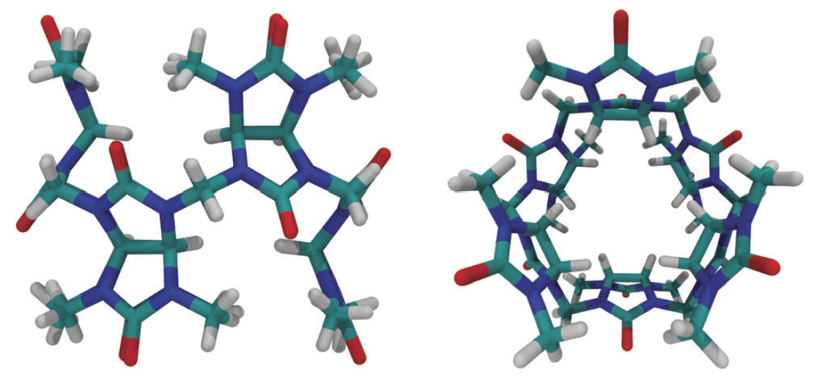
Fig. 1. Side and top views of Bambus[6]uril.
Reprint form: Chem. Soc. Rev., 2015, 44, 394 - Published by The Royal Society of Chemistry
Bambus[6]urils exhibit outstanding host-guest associated binding properties not only in aqueous media, but also in nonpolar solvents [2-4]. For example, Bambus[6]urils are able bind selectively different size anions in chloroform [5] and could be applied in the real-time analysis of anion mixtures by NMR methods [6]. In addition, the macrocycles are used as a carrier in tailor-made liquid membranes for highly selective electromembrane extractions of inorganic anions [7] and as a transmembrane Cl-/HCO3- antiporter [8].
References:
- Chem. Soc. Rev., 2015, 44, 394.
- Angew. Chem. Int. Ed. 2010, 49, 2378.
- Org. Lett., 2011, 13, 4000.
- Chem. Commun., 2014, 50, 1372.
- ChemPlusChem 2015, 80, 1601.
- Chem. Commun., 2015, 51, 4666.
- Analytica Chimica Acta 2017, 950, 49.
- Chem., 2019, 5, 429.
Featured Bambusurils:
07-1340 Dodecabenzylbambus[6]uril, Bn-BU[6] (1308315-95-8)
07-1342 Dodecamethylbambus[6]uril hydrate, BU[6] (1227292-62-7)