'Product Blog'
Iridium
The “Rainbow” Element
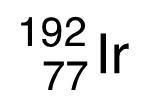
Browse the full iridium catalog here
Discovery – 1803, Smithson Tennant, England
Iridium was discovered sometime after its chemical neighbor, platinum (Pt). In 1748, platinum was discovered in Colombia.1 Platinum was analyzed by creating soluble salts using aqua regia (HCl/HNO3), but scientists also noted an insoluble black salt that was persistent in these studies.2 In 1799, French chemist Joseph Louis Proust labeled this insoluble black salt as “nothing less than graphite or plumbago.”2
Seeking to elucidate what this material was, Louis Nicolas Vauquelin treated this salt with acidic and basic solutions and obtained a volatile oxide.3 Vauquelin concluded a new metal must be present, which he named “ptene” (deriving from the Greek ptenos for “winged”). Following the work of Vauquelin, Smithson Tennant treated this insoluble black salt with NaOH followed by HCl and obtained dark, red crystals (thought to be of the formula Na2[IrCl6]·H2O). Upon heating these crystals, Tennant observed a powder which, “appeared of a white color, and was not capable of being melted, by any degree of heat I could apply…I should incline to call this metal Iridium, from the striking variety of colors which it gives, while dissolving in marine acid [HCl]…”5 The given name refers to Iris, the Greek goddess of rainbows, and the striking colors iridium salts can adopt.6
Fun Facts
- Second densest element (behind Os)
- Most corrosion-resistant metal
- An iridium-rich layer in the Earth’s crust helped give rise to the Alvarez hypothesis, that a massive extraterrestrial object was responsible for dinosaur extinction
- Crucibles made from iridium are used in growing high-quality single crystals
- Recently, iridium has seen use in photodynamic therapy for cancer
Iridium Products and Functions
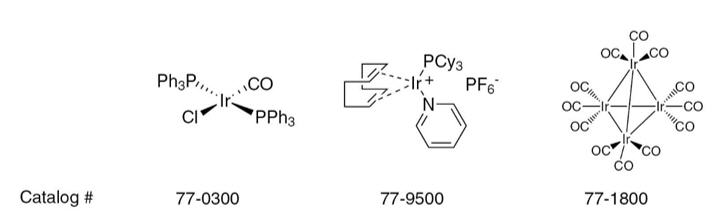
77-0300: Vaska’s complex. This complex can bind O2 reversibly and undergoes oxidative additions with electrophiles.
77-9500: Crabtree’s catalyst. An air-stable catalyst for hydrogenation and hydrogen-transfer reactions.
77-1800: Tetrairidium Dodecacarbonyl. A useful catalyst for the watershift gas reaction.
Browse the full iridium catalog here
References
- Relacíon histórica del viage a la América Meridional,1748.
- Platinum Metals Rev., 1987, 1, 32.
- Nature’s Building Blocks: An A-Z Guide to the Elements, 2011.
- Platinum Metals Rev.,2004, 48, 182.
- Phil. Trans., 1804, 94, 411.
- Greek Legends and Stories, 2000.