'Product Blog'
New Ligand Design Made Possible through Bis-oxazoline Ligands Modified with Side Arms (SaBOX)
Insertion of C2-Symmetry-Breaking Groups into Bis-oxazolines Improves the Enantioselectivty in Several Asymmetric Reactions
C2-symmetric bis-oxazolines currently dominate in asymmetric synthesis. However, introducing side arms (SA) into this symmetry may lead to the development of new ligand design. The installation of pendant C2-symmetry-breaking groups on oxazoline linkers resulted in a new type of ligand:
*Side arm bis-oxazolines (also known as SaBOX) with one or two pendant groups other than oxazoline at the bridge.
In comparison to their parent BOX ligands, the metal catalysts based on these SaBOX ligands usually exhibit a higher efficiency, and diastereo- and enantioselectivity with better impurity tolerance and stability.1
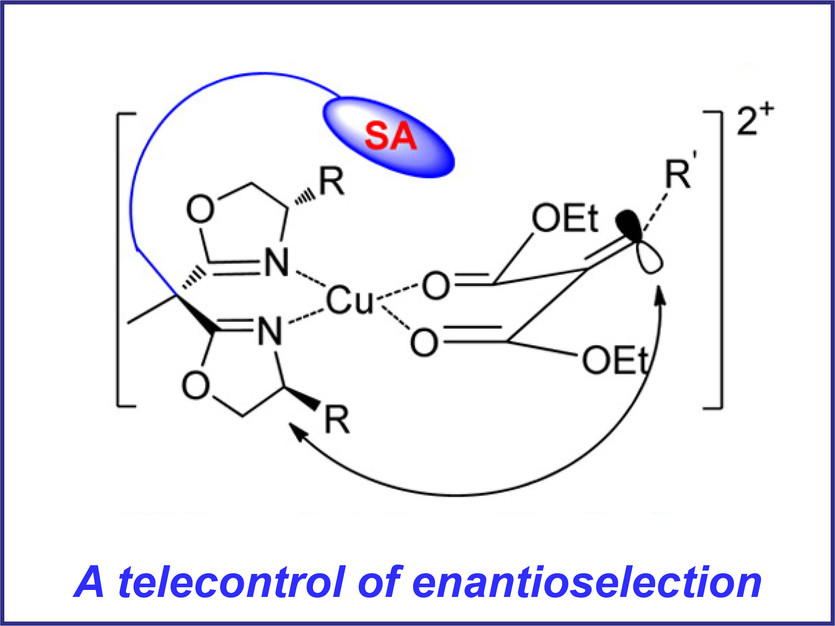
Figure 1. Relay the chirality by side arm group (SA)
Reprinted with permission from S. Liao et al, Acc. Chem. Res. 2014, 47, 2260-2272 [1].
Copyright (2014) American Chemical Society.
The SA group at the bridge carbon of the bis-oxazoline ligands can affect the electronic and steric properties of the catalytic site (Figure 1). The side arm can perform differently depending on the properties of the chosen groups. For example, the side arm could exert only a steric effect by virtue of its steric hindrance, or bind to the metal like a pendant ligand. In addition, , the side arm may behave as a directing group to assist in the stereochemical control and reagent/substrate activation.1
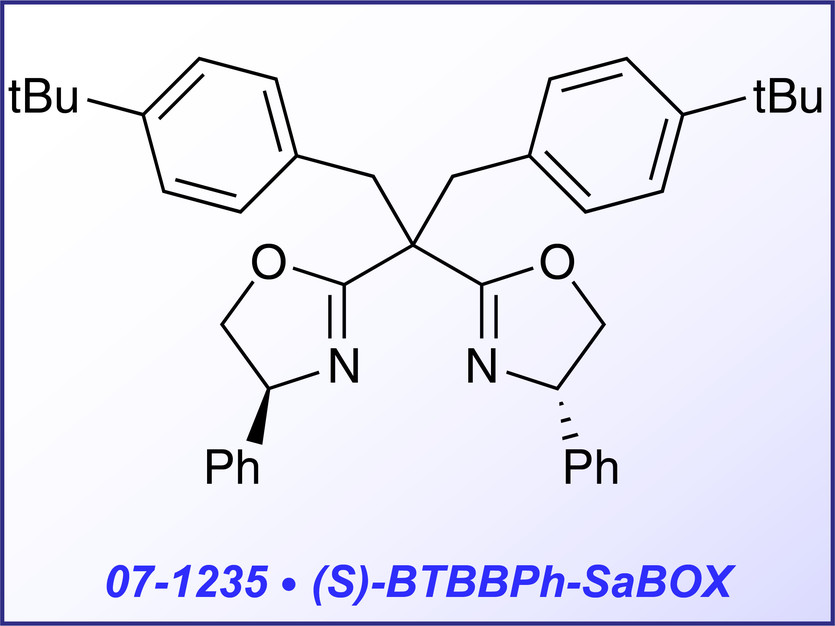
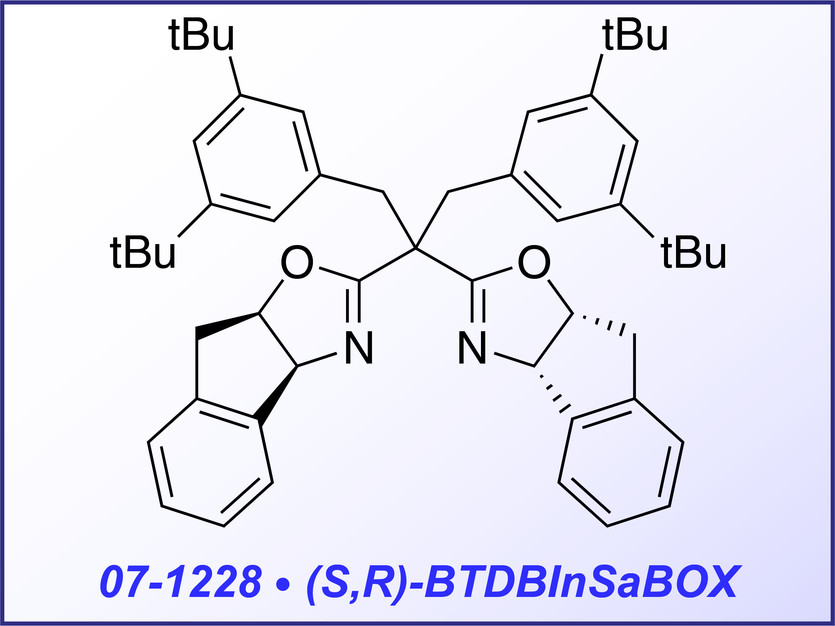
(S)-BTBBPh-SaBOX (07-1235) and (S,R)-BDTBIn-SaBOX (07-1228) represent double side armed bis-oxazolines, which are especially effective for copper- catalyzed cyclopropanation of multisubstituted olefins with phenyliodonium yilide malonate2 and cyclopentannulation of indoles with cyclopropanes.3 Both reactions are characterized with extremely high enantioselective yield (up to 99% and 96% respectively).
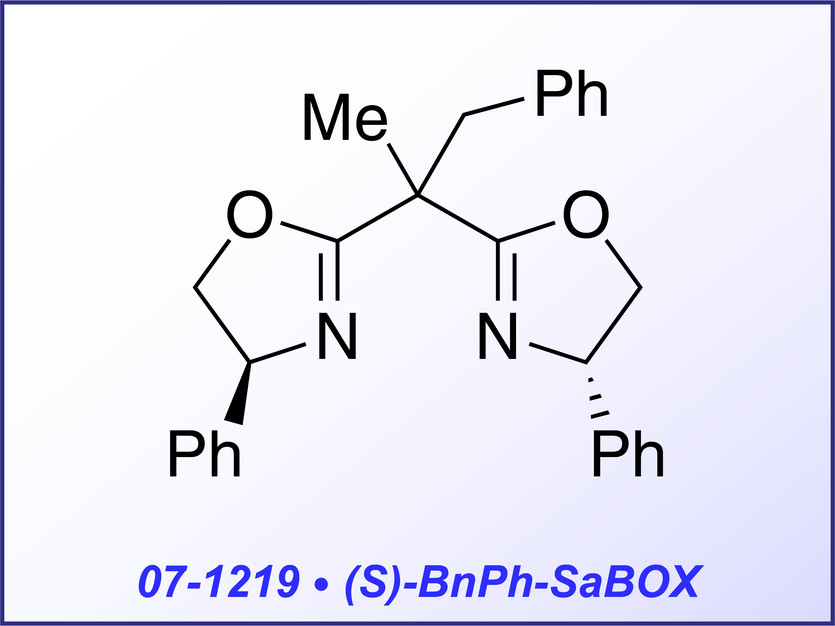
The mono side and arm-modified bis-oxazoline ligand (S)-BnPh-SaBOX (07-1219) is able to improve the reactivity and stereoselectivity in the asymmetric cyclopropanation of multisubstituted olefins with diazoacetates. Cis- and trans-1,2-substituted alkenes can be converted into the corresponding trisubstituted cyclopropanes, with high levels of diastereo- and enantioselectivity (>99:1 trans/cis and up to 98%).4
References:
- S. Liao et al, Acc. Chem. Res., 2014, 47, 2260-2272.
- C. Deng et al, Angew. Chem. Int. Ed., 2012, 51, 11620-11623.
- H. Xiong et al, J. Am. Chem. Soc., 2013, 135, 7851-7854.
- J. Li et al, Angew. Chem. Int. Ed., 2012, 51, 8838-8841.
Products mentioned in this blog:
Related Literature: