'Product Blog'
New Sun Light-Driven Water Splitting Catalysts for Hydrogen Production
Cobalt and Nickel Molecular Catalytic Sensitizers are Able to Catalyze Water Splitting Reactions Using Solar Energy
De-carbonization of the global energy systems is a major challenge. In response, renewable energy sources such as photovoltaic power, wind power or biomass conversions to carbon-free energy carriers continue to mature. In this respect, hydrogen produced from renewable resources is a promising potential source of clean energy. However, at the present time carbon-intensive fossil fuels are still the dominant source of industrial hydrogen.
Recently, Erwin Reisner and associates from the University of Cambridge developed new catalytic materials, which are based on inexpensive and widely available materials. These catalysts can efficiently affect water splitting via solar energy [1].
When semiconducting materials (e.g. TiO2), are co-functionalized with a dye (e.g. Ru) and catalyst, then exposed to light, the dye gets excited and oxidized. Generated electrons are then transferred through the conduction band of the semiconductor and are injected directly into a H2 evolution catalyst. The oxidized dye is regenerated by a water oxidation catalyst closing thus the fuel-making cycle (Fig. 1).
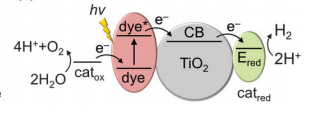
Fig. 1. Schematic representation of dye-sensitised photocatalysis (DSP) for water splitting.
catox = water oxidation catalyst, catred = H2 evolution catalyst [1]
©[Chem. Soc. Rev., 2015, DOI: 10.1039/c5cs00733j] - Published by The Royal Society of Chemistry
Chemical structures of cobalt [27-3015] and nickel [28-1720] water splitting catalysts are shown in figure 2. Triethylammonium [chlorobis(dimethylglyoximato)(4-hydrogenphosphonatepyridinyl) cobaltate(III)] [27-3015], also known as a cobaloxime (CoP), is an inexpensive catalyst that leads to the evolution of H2 either electro- or photocatalytically in pH-neutral water, even in the presence of atmospheric O2. The phosphonic acid group in (Et3NH)[CoP] enables the complex to dissolve in water and allows for its immobilization on metal oxide surfaces for heterogeneous applications [2].
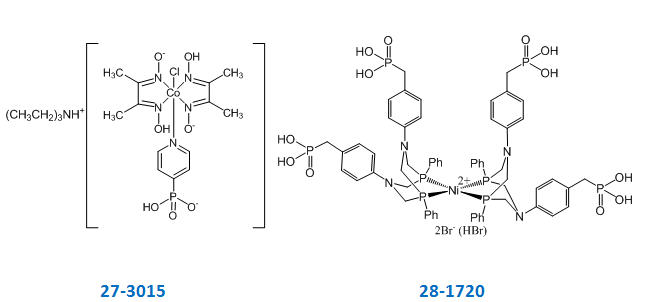
Fig. 2. Chemical structures of molecular cobalt [27-3015] and nickel [28-1720] water splitting catalysts
Bis{P,P'-1,5-diphenyl-3,7-bis[(4-hydrogenphosphonate)phenyl]-1,5,3,7-diazadiphosphocine}nickel(II) bromide (hydrogen bromide adduct) [28-1720], also known as a Ni bis(diphosphine)(NiP) catalyst, is an alternative water-splitting catalyst. The NiP inner core of the complex is responsible for catalytic activity. The phosphonic acid group in the outer-sphere provides the catalyst with good solubility in aqueous solutions. The product can also be anchored onto semiconductor particles [5].
As mentioned, the CoP catalyst shows a tolerance towards O2, but its activity suffers in the presence of CO. In contrast, the NiP catalyst is strongly inhibited by O2, but can produce H2 under high CO concentrations [6].
References:
- Chem. Soc. Rev., 2015, 45, 9.
- Angew. Chem. Int. Ed., 2012, 51, 9381.
- Angew. Chem. Int. Ed., 2012, 51, 12749.
- Chem. Commun., 2014, 50, 12768.
- J. Am. Chem. Soc., 2014, 136, 356.
- Chem. Commun., 2014, 50, 15995.
- Angew. Chem. Int. Ed., 2014, 53, 11538.
- J. Amer. Chem. Soc., 2015, 137, 6018.
Products mentioned in this blog:
Review our below literature sheet for even more product details:
Dye-Sensitised Semiconductors Modified with Molecular Catalysts for Light-Driven H2 Production