'Product Blog'
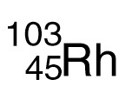
Rhodium
We offer over 100 rhodium products!
Discovery – 1803, William Hyde Wollaston, England
The discovery of rhodium was contemporary with Wollaston’s discovery of palladium.1 While working with a sample of crude platinum ore from South America,2 Wollaston sought to extract the individual metals present. The primary component of this ore, platinum, had been isolated and discovered half a century earlier,3 but certain residues from this crude sample would not dissolve in aqua regia (3:1 hydrochloric:nitric acids) and were not yet identified.
Wollaston’s process began with treating the crude ore with aqua regia, followed by neutralization with NaOH. Addition of NH4Cl afforded ammonium chloroplatinate, which had already been discovered. Wollaston knew from his previous work that addition of dilute HNO3 to the remaining dissolved ore would afford palladium. With platinum and palladium removed from the crude sample, sodium chloride was added, and a rose‐red precipitate was formed.
This precipitate was reconstituted in ethanol and treated with zinc, which displaced the central metal in the rose‐red precipitate to afford pure rhodium metal.1
Fun Facts
- Rhodium sees widespread use in three‐way catalytic converters to reduce emissions in automobile exhaust7,8
- The name rhodium comes from the Greek word “rhodon” meaning rose‐colored, a reference to the color of dilute solutions of rhodiumsalts
- Rhodium is highly corrosion‐ and acid‐resistant – even aqua regia cannot dissolve rhodium
- While rhodium is a fission product of 235U, the process of extracting rhodium from used nuclear waste has not yet been accomplished, due to high costs and safetyissues4
Rhodium Products and Functions
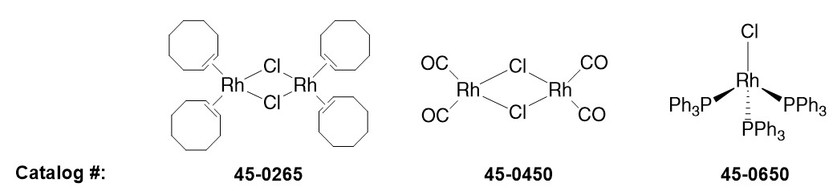
45‐0265: Chlorobis(cyclooctene)rhodium(I) dimer. Rhodium with easily displaced ligands, which have been used to make chiral catalysts for asymmetric alkene hydrogenation.5
45‐0450: Chlorodicarbonylrhodium(I) dimer. A catalyst used in rhodium‐catalyzed Pauson‐Khand reactions.6
45‐0650: Wilkinson’s Catalyst. Used in the hydrogenation of alkenes.
References:
- Platinum Metals Rev., 2003, 47,175.
- CRC Handbook of Chemistry and Physics: A Ready‐Reference Book of Chemical and Physical Data, 2004, 4.
- Phil. Trans. 1749, 46, 584.
- Platinum Metals Rev., 2005, 49, 79.
- Adv. Synth. Catal., 2003, 345, 3.
- J. Organomet. Chem., 2001, 624, 73
- Chemical Engineering Journal. 82 (1–3): 149–156.
- Applied Catalysis A: General. 221 (1–2): 443–457.