'Product Blog'
AntPhos Ligands for Efficient Cross Coupling and Asymmetric Cyclization
The recently developed AntPhos ligand provides an attractive platform for metal-catalyzed cross-coupling and enantioselective cyclization reactions
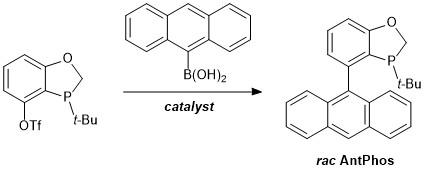
The AntPhos ligand class is derived from a five-membered heterocycle containing both phosphorus and oxygen fused with an aromatic ring containing a pendant anthracene group. Introduced by Dr. Wenjun Tang and co-workers at Boehringer Ingelheim in 2011,1 these ligands are readily synthesized by cross-coupling of the parent aryl triflate with an arylboronic acid, the same route by which the structurally related BI-DIME ligands are synthesized.
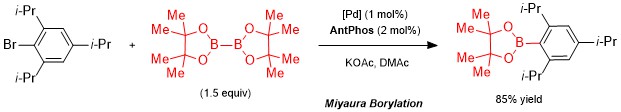
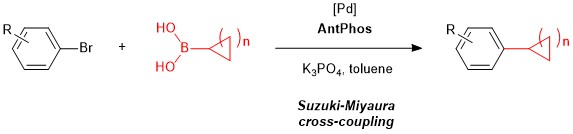
AntPhos has seen application as a ligand in a variety of cross-coupling reactions such as Pd-catalyzed Miyaura Borylation1 and Suzuki-Miyaura cross-coupling2 reactions using a variety of coupling partners. Though these transformations can be accomplished by a variety of catalysts, an area where AntPhos distinguishes itself is in its ability to catalyze the cross-coupling of sterically hindered substrates, such as 2,6-disubstituted aryl bromides with cyclic alkylboronic acids. These substrate pairings, especially secondary alkylboronic acids, were previously inaccessible by other catalyst systems, and the development of the AntPhos ligands has helped to further expand the scope of coupling partners in these reactions.
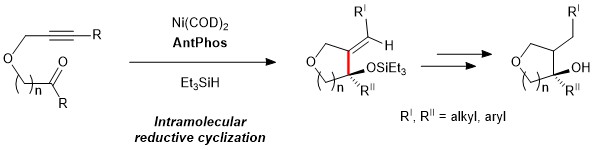
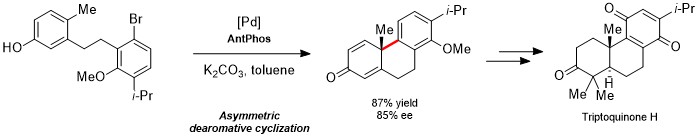
Due to the rigid conformation of the phosphorus atom, the enantiomers of the AntPhos ligand are able to be resolved and isolated, opening up the potential to use these ligands in asymmetric transformations. Recently, the chiral versions of AntPhos have been leveraged in Nickel-Catalyzed intramolecular reductive cyclizations of alkenones, allowing for the formation of chiral products in exceptionally high enantiomeric excess of up to 99%.3 Another recent application of the AntPhos ligand is found in Pd-catalyzed asymmetric dearomative cyclization to form tricyclic fused ring compounds, enabling the synthesis of a key intermediate towards the formation of Triptoquinone H in 87% yield and 85% ee.4
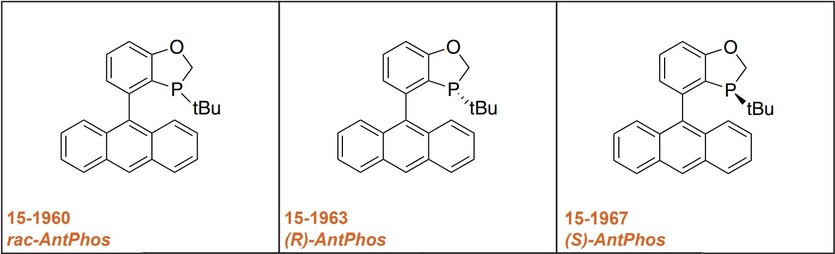
Strem offers both the racemic mixture as well as the resolved enantiomers of the AntPhos ligand.
References
1. Org. Lett. 2011, 13, 1366.
2. Org. Chem. Front., 2014, 1, 225-229.
3. Angew. Chem. Int. Ed., 2015, 54, 2520.
4. Tetrahedron, 2016, 72, 1782.
Products mentioned in this blog can be found on our literature sheet: